Richard Dorsky
Professor of Neurobiology
Developmental Neurobiology

Molecular Biology Program
Education
B.A. University of California, Berkeley
Ph.D. University of California, San Diego
Research
During development of the central nervous system (CNS), cells are influenced by their environment to adopt specific fates. These environmental signals are crucial for the correct patterning of cell types and their subsequent functional connections. My laboratory is studying the role of the Wnt/β-catenin signaling pathway in CNS cell fate specification. We use zebrafish as a model organism, focusing on the regulation of Wnt/β-catenin target genes in the CNS by a family of transcription factors called Tcf proteins. Tcfs are required for β-catenin to regulate transcription, and can act as repressors or activators of target genes depending on the state of Wnt signaling.
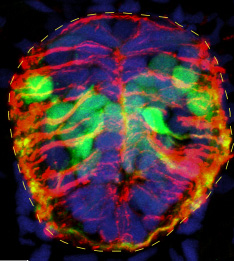
Spinal Cord Neurogenesis
We are interested in the functions of Wnt signaling in zebrafish spinal cord development and regeneration. We have shown that following transection of the larval spinal cord, Wnt signaling is required for radial glia to generate new neurons. One current project in the lab is determining the mechanism downstream of Wnt signaling that mediates neurogenesis following injury. This function may be critical for stem cell-mediated regeneration. A second project is investigating whether new neurons integrate into functional circuitry, and whether they are required for the recovery of sensory and motor behavior.
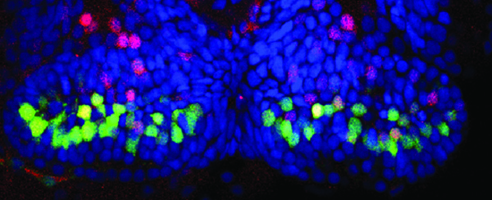
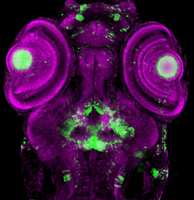
Hypothalamic Neurogenesis
This work focuses on the role of Wnt signaling and its transcriptional mediator Lef1 in the differentiation of neural progenitors in the posterior hypothalamus. This region of the brain maintains Wnt activity and continues to produce neurons throughout life, suggesting that Wnt-regulated neurogenesis plays an important role in the adult brain. We have found an evolutionarily conserved requirement for Lef1 in mediating anxiety, and in the formation of anxiolytic hypothalamic neurons. We have also identified Lef1 target genes in the zebrafish and mouse hypothalamus, and are determining their functions in neurogenesis and behavior. Other work is focused on testing the requirement for postembryonic neurogenesis in modulating anxiety.
References
- , , , , , , , otpb functions in a Lef1-dependent transcriptional network required for expression of the stress response inhibitor crhbp in the zebrafish hypothalamus.
- Unique advantages of zebrafish larvae as a model for spinal cord regeneration. Front Mol Neurosci. 2022;15:983336. doi: 10.3389/fnmol.2022.983336. eCollection 2022.. PubMed PMID: 36157068; PubMed Central PMCID: PMC9489991.
- Vasudevan D, Liu YC, Barrios JP, Wheeler MK, Douglass AD, Dorsky RI. Regenerated interneurons integrate into locomotor circuitry following spinal cord injury. Exp Neurol. 342 (2021):113737.
- Hutcheson DA, Xie Y, Figueroa P, Dorsky RI. A transgene targeted to the zebrafish nkx2.4b locus drives specific green fluorescent protein expression and disrupts thyroid development. Dev Dyn 249(11) (2020):1387-1393.
- Xie Y, Dorsky RI. Development of the hypothalamus: conservation, modification and innovation. Development 144 (2017), 1588-1599.
- Xie Y, Kaufmann D, Moulton MJ, Panahi S, Gaynes JA, Watters HN, Zhou D, Xue HH, Fung CM, Levine EM, Letsou A, Brennan KC, Dorsky RI. Lef1-dependent hypothalamic neurogenesis inhibits anxiety. PLoS Biology 15, e2002257.
- McPherson AD, Barrios JP, Luks-Morgan SJ, Manfredi JP, Bonkowsky JL, Douglass AD, Dorsky RI. Motor Behavior Mediated by Continuously Generated Dopaminergic Neurons in the Zebrafish Hypothalamus Recovers after Cell Ablation. Current Biology 26 (2016) 263-9.
- Duncan RN, Xie Y, McPherson AD, Taibi AV, Bonkowsky JL, Douglass AD, Dorsky RI. Hypothalamic radial glia function as self-renewing neural progenitors in the absence of Wnt/ß-catenin signaling. Development 143 (2016) 45-53.
- Briona LK, Poulain FE, Mosimann C, Dorsky RI. Wnt/ß-catenin signaling is required for radial glial neurogenesis following spinal cord injury. Developmental Biology 403 (2015) 15-21.
- Otsuna H, Hutcheson DA, Duncan RN, McPherson AD, Scoresby AN, et al. High-resolution analysis of central nervous system expression patterns in zebrafish Gal4 enhancer-trap lines. Developmental dynamics : an official publication of the American Association of Anatomists 244 (2015) 785-96.
- Briona LK and Dorsky RI. Radial glial progenitors repair the zebrafish spinal cord following transection. Experimental Neurology 256 (2014) 81-92.
- Wang X, Kopinke D, Lin J, McPherson AD, Duncan RN, Otsuna H, Moro E, Hoshijima K, Grunwald DJ, Argenton F, Chien CB, Murtaugh LC and Dorsky RI. Wnt Signaling Regulates Postembryonic Hypothalamic Progenitor Differentiation. Developmental Cell 23 (2012) 624-636.
- Bonner J, Letko M, Nikolaus OB, Krug L, Cooper A, Chadwick B, Conklin P, Lim A, Chien CB and Dorsky RI. Midline crossing is not required for subsequent pathfinding decisions in commissural neurons. Neural Development 7 (2012) 18.
- Kim HS and Dorsky RI. Tcf7l1 is required for spinal cord progenitor maintenance. Developmental Dynamics 240 (2011) 2256-64.
- Lin J, Wang X and Dorsky RI. Progenitor expansion in apc mutants is mediated by Jak/Stat signaling. BMC Developmental Biology 11 (2011) 73.
- Gribble SL, Kim HS, Bonner J, Wang X and Dorsky RI. Tcf3 inhibits spinal cord neurogenesis by regulating sox4a expression. Development 136 (2009) 781-9.
- Wang X, Lee JE and Dorsky RI. Identification of Wnt-responsive cells in the zebrafish hypothalamus. Zebrafish 6 (2009) 49-58.
- Veien ES, Rosenthal JS, Kruse-Bend RC, Chien CB and Dorsky RI. Canonical Wnt signaling is required for the maintenance of dorsal retinal identity. Development 135 (2008) 4101-11.
- Bonner, J, Gribble SL, Veien ES, Nikolaus OB, Weidinger G and Dorsky RI. Distinct pathways mediate patterning and proliferation in the dorsal spinal cord downstream of canonical Wnt signaling. Developmental Biology 313 (2008) 398-407.
- Gribble SL, Nikolaus OB and Dorsky RI. Regulation and function of Dbx genes in the zebrafish spinal cord. Developmental Dynamics 236 (2007) 3472-83.
- Nyholm MK, Wu S, Dorsky RI and Grinblat Y. The zebrafish zic2a-zic5 gene pair acts downstream of canonical Wnt signaling to control cell proliferation in the developing tectum. Development 134 (2007) 735-746.
- Lee J, Wu S, Goering LM and Dorsky RI. Canonical Wnt signaling through Lef1 is required for hypothalamic neurogenesis. Development 133 (2006) 4451-4461.
- Veien ES, Grierson MJ, Saund RS and Dorsky RI. Expression pattern of zebrafish tcf7 suggests unexplored domains of Wnt/beta-catenin activity. Developmental Dynamics 233 (2005) 233-239.
- Lewis JL, Bonner J, Modrell M, Ragland JW, Moon RT, Dorsky RI and Raible DW. Reiterated Wnt signaling during zebrafish neural crest development. Development 131 (2004) 1299-1308.
- Dorsky RI, Itoh M, Moon RT and Chitnis A. Two tcf3 genes cooperate to pattern the zebrafish brain. Development 130 (2003) 1937-1947.
- Dorsky RI, Sheldahl LC and Moon RT. A Transgenic Lef1/β-catenin-Dependent Reporter is Expressed in Spatially -Restricted Domains Throughout Zebrafish Development. Developmental Biology 241 (2002) 229-237.
- Dorsky RI, Raible DW and Moon RT. Signaling pathways and neural crest fate specification. BioEssays 22 (2000) 708-716.
