Andres Villu Maricq
Professor of Neurobiology and Adjunct Professor of Biological Sciences
Mechanisms and Development of Synaptic Function, Gluamate Receptors, Neural Circuits

Molecular Biology Program
Education
B.S. Brown University
M.D. University of California, San Francisco
Ph.D. University of California, Berkeley
Research
Identification and Characterization of Auxiliary Proteins that Contribute to Synaptic Function
Our laboratory has developed new genetic strategies to uncover the molecular machinery required for synaptic transmission. In a series of studies in the nematode C. elegans, we have identified new classes of auxiliary subunits that contribute to AMPAR function, shown that they have dramatic effects on in vivo glutamate-gated currents, and demonstrated that mutations in these genes predictably modify specific behaviors.
The Delivery, Removal and Redistribution of Synaptic AMPA Receptors
Using an in vivo approach in C. elegans, we recently demonstrated the central importance of UNC-116, the homolog of vertebrate kinesin-1 heavy chain (KIF5), for the delivery, and surprisingly, the removal and redistribution of synaptic AMPARs. We show that UNC-116/KIF5 motors provide a rapid-response mechanism for the precise regulation of the number of AMPARs at synapses – a major determinant of synaptic strength.
Translocation of Synaptic Receptors
We also study how synaptic receptors are delivered from subsynaptic stores to the surface of the synapse. We found that Wnt molecules, evolutionarily conserved secreted glycoproteins that are best known for their roles in early development, also have an ongoing role in the adult nervous system to control translocation of the ACR-16/a7 nicotinic receptors and change the strength of synaptic transmission.
The Control of Behavior
We have an ongoing interest in understanding the molecular basis of well-defined quantifiable behaviors. Early efforts were directed at the control of rhythmic behaviors in C. elegans. More recently, we have initiated mechanistic studies of a neural circuit that contributes to navigation along gradients of sensory information.
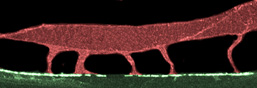
False-color confocal image of C. elegans body wall muscle. Muscle arms emerge from the cell body and extend downwards, making contact with nerves at the tips
References
Identification and Characterization of Auxiliary Proteins that Contribute to Synaptic Function
- Brockie, P.J., M. Jensen, J.E. Mellem, E. Jensen, T. Yamasaki, R. Wang, D. Maxfield, C. Thacker, F. Hoerndli, P.J. Dunn, S. Tomita, D.M. Madsen and A.V. Maricq (2013) Cornichons control ER export of AMPA receptors to regulate synaptic excitability. Neuron 80:129-42.
- Wang, R., J.E. Mellem, M. Jensen, P.J. Brockie, C.S. Walker, F.J. Hoerndli, D.M. Madsen and A.V. Maricq (2012) The SOL-2/Neto auxiliary protein modulates the function of AMPA-subtype ionotropic glutamate receptors. Neuron 75:838-50.
- Wang, R., C.S. Walker, P.J. Brockie, M.M. Francis, J.E. Mellem, D.M. Madsen and A.V. Maricq (2008) Evolutionary conserved role for TARPs in the gating of glutamate receptors and tuning of synaptic function. Neuron 59:997-1008.
- Zheng, Y., J.E. Mellem, P.J. Brockie, D.M. Madsen and A.V. Maricq (2004) SOL‑1 is a CUB-domain protein required for GLR‑1 glutamate receptor function in C. elegans. Nature 427:451-57.
The Delivery, Removal and Redistribution of Synaptic AMPA Receptors
- Hoerndli, F., R. Wang, J. Mellem, A. Kallarackal, P. Brockie, C. Thacker, D. Madsen and A.V. Maricq (2015) Neuronal activity and CaMKII regulate kinesin-mediated transport of synaptic AMPARs. Neuron 86:457-74. (NIHMSID: 670172)
- Hoerndli, F.J., D.A. Maxfield, P.J. Brockie, J.E. Mellem, E. Jensen, R. Wang, D.M. Madsen and A.V. Maricq (2013) Kinesin-1 regulates synaptic strength by mediating the delivery, removal and redistribution of AMPA receptors. Neuron 80:1421-37. [Accompanying Neuron Preview: Rongo, C. (2013) Going mobile: AMPA receptors move synapse to synapse in vivo, 80: 1339-41]
- Video abstract http://www.sciencedirect.com/science/article/pii/S0896627313010027
Translocation of Synaptic Receptors
- Jensen, M., F.J. Hoerndli, P.J. Brockie, R. Wang, E. Johnson, D. Maxfield, M.M. Francis, D.M. Madsen and A.V. Maricq (2012) Wnt signaling regulates acetylcholine receptor translocation and synaptic plasticity in the adult nervous system. Cell 149:173-87.
- Francis, M.M., S.P. Evans, M. Jensen, D.M. Madsen, J. Mancusco, K. Norman and A.V. Maricq (2005) The Ror receptor tyrosine kinase CAM-1 is required for ACR-16 mediated synaptic transmission at the C. elegans neuromuscular junction. Neuron 46:581-94.
The Control of Behavior
- Kano, T., P.J. Brockie, T. Sassa, H. Fujimoto, Y. Kawahara, Y. Iino, J.E. Mellem, D.M. Madsen, R. Hosono and A.V. Maricq (2008) Memory in Caenorhabditis elegans is mediated by NMDA-type ionotropic glutamate receptors. Curr. Biol. 18:1010-15.
- Norman, K.R., R.T. Fazzio, J.E. Mellem, M.V. Espelt, K. Strange, M. Beckerle and A.V. Maricq (2005) The Rho/Rac-family guanine nucleotide exchange factor VAV-1 regulates rhythmic behaviors in C. elegans. Cell 123:119-32.
