Yukio Saijoh
Adjunct Assistant Professor of Neurobiology
Left-Right Asymmetry, Heart Development

Molecular Biology Program
Education
Research
My fundamental interest is in vertebrate pattern formation: how vertebrates make up
their complicated and organized body plan. Embryogenesis requires correct cell differentiation
and positioning of tissues and organs in three dimensions. These two events are closely
related to each other and are regulated with respect to each of the three body axes:
anterior-posterior, dorso-ventral, and left-right (LR). Defects in LR axis information
result in disorganized morphology and malpositioning of organs that disrupt their
normal function. In humans, defects in LR asymmetric patterning commonly cause birth
defects of the heart, lungs, gut and other asymmetric organs. LR asymmetric morphogenesis
is regulated by LR axis information established during the early somite stages of
development. The genetic cascade for establishing initial LR asymmetry has been identified
over the last 10 years, but the subsequent steps by which LR information regulates
asymmetric morphogenesis of internal organs is poorly understood. The goal of my research
is to understand the genetic cascade critical for establishing LR asymmetries that
is observed in the internal organs of vertebrates such as the heart, stomach and intestines.
Our current emphasis is on studying how LR asymmetry is established in the lateral
plate mesoderm that is precursor of internal organs, and how the signals then regulate
LR asymmetric morphogenesis in internal organs.
First, we are focusing on the roles of endoderm in establishing LR asymmetry. We have
studied mouse Sox17 mutants that have specific initial defects in endoderm cells.
This study has revealed that endoderm is important for establishing LR signals before
Nodal expression in the lateral plate. We are analyzing molecular mechanisms by which
LR asymmetry is regulated by the endoderm cell population.
Second, we are investigating how LR signals regulate asymmetric morphogenesis in heart
looping. Cardiac cells derived from the left and right cardiac fields fuse to form
a single heart tube and then the tube loops rightward. During heart tube formation
and looping, cardiac cells differentiate to start beating. To understand basics of
dynamic morphogenesis, we are examining cell behavior during heart tube formation
and looping such as cell movement, cell proliferation and cell polarity in the chick
system, which is an ideal model to observe heart development in vivo and in culture.
Information derived from studies in chicks is used to understand heart looping in
mammalian systems, such as the mouse with its valuable genetic tools.
Third, we are interested in intestinal asymmetric morphogenesis using mouse models
of human intestine disorders such as malrotation with the same approach as above.
Interestingly, intestines develop outside of embryos. The intestines then return into
the body just before birth synchronizing with body-wall closure completion. If the
timing of these two events is different, intestines are left outside of the body,
which causes a congenital disease called omphalocele. Thus, we are also investigating
body-wall closure morphogenesis as a model for human disease ompholocele.
These phenomena in internal organogenesis share the same basic cellular events such
as cell proliferation, cell migration, cell to cell adhesion and communication, and
cell differentiation. I take several approaches using different model systems to reveal
the morphogenesis of LR asymmetry in internal organs.
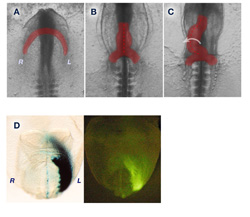
Legend: (A-C) Heart looping morphogenesis in the chick embryo. (A) Left and right cardiac myocytes starts to fuse at the anterior region at stage 8. (B) Fused linear heart tube just before looping at stage 10. (C) The heart tube is looping toward the right side at stage 11. (D) Left sided LacZ reporter expression driven by the asymmetric enhancer of Lefty2. Expression includes a region of the fusing heart in mouse embryos 8 days post coitum.
References
-
H Kidokoro, S. Yonei-Tamura, K. Tamura, G. C. Schoenwolf, Y. Saijoh (2018). The Heart Tube Forms and Elongates Through Dynamic Cell Rearrangement Coordinated with Foregut Extension. Development. 145(7). pii: dev152488. doi: 10.1242/dev.152488.
-
Chua J.S., Tran V.M., Kalita M., Quintero M.V., Antelope O., Muruganandam G., Saijoh Y.*, Kuberan B* (2017) A glycan-based approach to therapeutic angiogenesis. PLoS One. 12(8): e0182301 *equal contribution
-
Higashiyama H, Ozawa A, Sumitomo H, Uemura M, Fujino K, Igarashi H, Imaimatsu K, Tsunekawa N, Hirate Y, Kurohmaru M, Saijoh Y, Kanai-Azuma M, Kanai Y. (2017) Embryonic cholecystitis and defective gallbladder contraction in the Sox17-haploinsufficient mouse model of biliary atresia. Development. 15;144(10):1906-1917.
- Saijoh, Y., Viotti, M. and Hadjantonakis, A.K. “Follow your gut: relaying information from the site of left-right symmetry breaking in the mouse.” Genesis, 2014. 52: 503-514.
- Hou, J., Wei, W., Saund, R.S., Xiang, P., Cunningham, T.J., Yi, Y., Alder, O., Lu, D.Y., Savory, J.G., Krentz, N.A., Montpetit, R., Cullum, R., Hofs, N., Lohnes, D., Humphries, R.K., Yamanaka, Y., Duester, G., Saijoh, Y. and Hoodless, P.A. “A regulatory network controls nephrocan expression and midgut patterning.” Development, 2014. 141: 3772-3781.
- Uemura, M., Ozawa, A., Nagata, T., Kurasawa, K., Tsunekawa, N., Nobuhisa, I., Taga, T., Hara, K., Kudo, A., Kawakami, H., Saijoh, Y., Kurohmaru, M., Kanai-Azuma, M. and Kanai, Y. “Sox17 haploinsufficiency results in perinatal biliary atresia and hepatitis in C57BL6 background mice.” Development, 2013. 140: 639-48.
- Kosaka, Y., Cieslik, K. A., Li, L., Lezin, G., Maguire, C.T., Saijoh,Y., Toyo-oka, K., Gambello, Ml. J., Vatta, M., Wynshaw-Boris, A., Baldini, A., Yost, H. J. and Brunelli, L. “14-3-3ε has a role in cardiac ventricular compaction by regulating the cardiomyocyte cell cycle.” Mol Cell Biol, 2012. 32: 5089-2102.
- Nichol, P.F., Corliss, R., Yamada, S., Shiota, K. and Saijoh, Y. “Muscle patterning in mouse and human abdominal wall development and ompahlocele specimens of humans.” Anat Rec, 2012. 295(12): 2129-40.
- Saund, R.S., Kanai-Azuma, M., Kanai, Y., Kim, I., Lucero, M.T. and Saijoh, Y. “Gut endoderm is involved in the transfer of left-right asymmetry from the node to the lateral plate mesoderm in the mouse embryo.” Development, 2012. 139 (13): p. 2426-35.
- Nichol, P.F., Tyrrell, J.D. and Saijoh, Y. “Retinaldehyde dehydrogenase 2 is down-regulated during duodenal atresia formation in Fgfr2IIIb-/- mice.” J Surg Res, 2012. 175(1): p. 82-7.
- Nichol, P.F. and Saijoh, Y. “Pitx2 is a critical early regulatory gene in normal cecal development.” J Surg Res, 2011. 170(1):107-11.
- Kwan, K.M., Otsuna, H., Kidokoro, H., Carney, K.R., Saijoh, Y. and Chien, C.B. “A complex choreography of cell movements shapes the vertebrate eye.” Development, 2012. 139(2): p. 359-72.
- Nichol, P.F., Botham, R., Saijoh, Y., Reeder, A.L. and Zaremba, K.M. “A more efficient method to generate null mutants using Hprt-Cre with floxed alleles.” J Pediatr Surg, 2011. 46(9): p. 1711-9.
- Nichol, P.F., Corliss, R.F., Tyrrell, J.D., Graham, B., Reeder, A. and Saijoh, Y. “Conditional mutation of fibroblast growth factor receptors 1 and 2 results in an omphalocele in mice associated with disruptions in ventral body wall muscle formation.” J Pediatr Surg, 2011. 46(1): p. 90-6.
- Kawasumi, A., Nakamura, T., Iwai, N., Yashiro, K., Saijoh, Y., Belo, J.A., Shiratori, H. and Hamada, H. “Left-right asymmetry in the level of active Nodal protein produced in the node is translated into left-right asymmetry in the lateral plate of mouse embryos.” Dev Biol, 2011. 353(2): p. 321-30.
- Nichol, P.F., Corliss, R.F., Tyrrell, J.D., Graham, B., Reeder, A. and Saijoh, Y. “Conditional mutation of fibroblast growth factor receptors 1 and 2 results in an omphalocele in mice associated with disruptions in ventral body wall muscle formation.” J Pediatr Surg, 2011. 46(1): p. 90-6.
- Bleyl, S.B., Saijoh, Y., Bax, A.M., Gittenberger-de Groot, A.C., Wisse, L.J., Chapman, S.C., Hunter, J., Shiratori, H., Hamada, H., Shiota, K., Klewer, S.E., Leppert, M.F. and Schoenwolf, G.C. “Dysregulation of the PDGF RA gene causes inflow tract anomalies including TAPVR: Integrating evidence from human genetics and model organisms.” Hum Mol Genet, 2010. 19(7): 1286-1301.
- Yun, S., Saijoh, Y., Hirokawa, K.E., Kopinke, D., Murtaugh, C.L., Monuki, E.S. and Levine, E.M. “Lhx2 links the intrinsic and extrinsic factors controlling optic cup formation.” Development, 2009. 36: 3895-3906.
- Park, E.J., Sun, X., Nichol, P., Saijoh, Y., Martin, J.F. and Moon, A.M. “System for tamoxifen-inducible expression of cre-recombinase from the Foxa2 locus in mice.” Dev Dyn, 2008. 237(2): 447-53.
- Kelly, G.M., Saijoh, Y., Finkielsztein, A. and Mangos, S. “Mouse G-protein gamma3 expression in the developing CNS and neural crest cell derivatives.” Int J Dev Biol, 2008. 52(8): 1143-50.
- Tsunoda, I., Tanaka, T., Saijoh, Y. and Fujinami, R.S. “Targeting inflammatory demyelinating lesions to sites of wallerian degeneration.” Am J Pathol, 2007. 171(5): 1563-75.
- Tanaka, C., Sakuma, R., Nakamura, T., Hamada, H. and Saijoh, Y. “Long-range action of Nodal requires interaction with GDF1,.” Genes Dev, 2007. 21(24): 3272-82.
- Sakamoto, Y., Hara, K., Kanai-Azuma, M., Matsui, T., Miura, Y., Tsunekawa, N., Kurohmaru, M., Saijoh, Y., Koopman, P. and Kanai, Y. “Redundant roles of Sox17 and Sox18 in early cardiovascular development of mouse embryos.” Biochem Biophys Res Commun, 2007. 360(3): 539-44.
- Oki, S., Hashimoto, R., Okui, Y., Shen, M.M., Mekada, E., Otani, H., Saijoh, Y. and Hamada, H. “Sulfated glycosaminoglycans are necessary for Nodal signal transmission from the node to the left lateral plate in the mouse embryo.” Development, 2007. 134(21): 3893-904.
- Frank, D.U., Elliott, S.A., Park, E.J., Hammond, J., Saijoh, Y. and Moon, A.M. “System for inducible expression of cre-recombinase from the Foxa2 locus in endoderm, notochord, and floor plate.” Dev Dyn, 2007. 236(4): 1085-92.
