Marina Venero Galanternik
Assistant Professor of Human Genetics
Meninges, Zebrafish, Developmental Biology, Meningeal Cells, Perivascular Meningeal Cells, Lymphatic System, Lymphatic Vessels, Meningeal Layers, High-resolution Microscopy

Molecular Biology Program
Education
B.S. Universidad Nacional Agraria La Molina
Ph.D. University of Utah
Research
Mechanisms regulating Meningeal Development
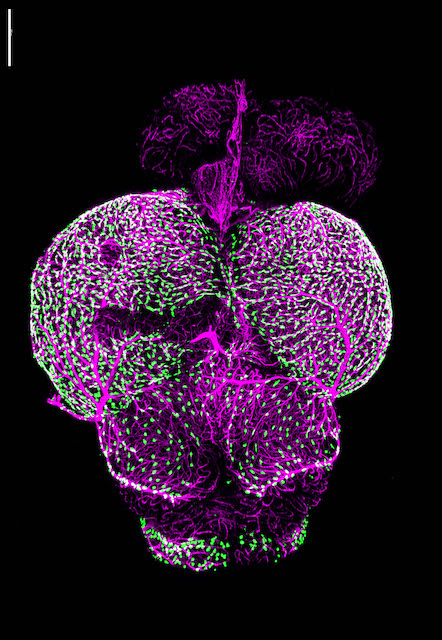
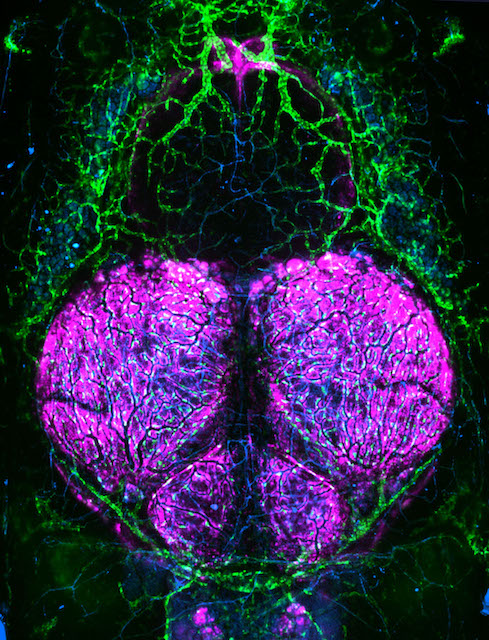
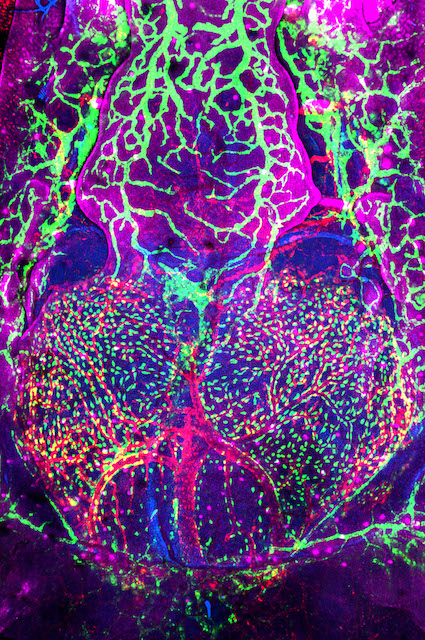
Research in the Venero Galanternik lab focuses on the meninges, an extremely important but poorly understood set of membranous tissues that envelop and protect the vertebrate central nervous system (CNS). The meninges are essential for waste removal from the brain and brain homeostasis, and they protect the CNS from mechanical insults and infection. Meningeal dysfunction due to disease is both highly prevalent and associated with high levels of morbidity and mortality. It is estimated that nearly 3 million cases of meningitis occur globally each year, causing the death of approximately 30% of these patients, and survivors are frequently left with permanent neurological damage. Similarly, pathological meningeal waste accumulation associated with a variety of chronic diseases and with aging is highly associated with neurodegeneration and neurocognitive decline. Despite their critical roles, little is known about resident meningeal cell populations and how they work together to accomplish their important protective functions.
In mammals the meninges lie immediately adjacent to a thick opaque skull, making them challenging to image and study and therefore, there has been limited characterization of meningeal cell types in mammals, and few specific markers and genes associated with these cell types have been identified. The genetic and experimental accessibility of the zebrafish, combined with the ability to perform high-resolution optical imaging of the brain surface through the thin, transparent skull of developing and even adult animals, make zebrafish an ideal research organism for studying the meninges. Our studies have revealed that, contrary to classical descriptions of teleost fishes, zebrafish have complex, multilayered meninges that strongly resemble those of mammals, making the fish a powerful and translatable model for comparative studies of meningeal development, function, and pathology. Our lab has established a strong anatomical, cellular and molecular foundation for zebrafish meningeal research. Our ongoing goals are focused on the understanding of the biological events regulating meningeal formation, meningeal cells functions and investigating the potential crosstalk between the meninges and their adjacent tissues during homeostasis and disease.
References
- Castranova D., Samasa B., Venero Galanternik M., Gore AV., Goldstein AE., Park JS. and Weinstein BM (2022). Long-term Imaging of Living Adult Zebrafish. https://doi.org/10.1242/dev.199667 - Development 149(4):dev199667.
- Stratman AN., Burns MC., Farrelly OM., Davis AE., Li W., Pham VN., Castranova D., Yano JJ., Goddard LM., Nguyen O., VeneroGalanternik M., Bolan TJ., Kahn ML., Mukouyama Y., Weinstein BM. (2021). Chemokine mediated signalling within arteries promotes vascular smooth muscle cell recruitment. Commun Biol3, doi.org/10.1038/s42003-020-01462-7
- Castranova D., Samasa B., Venero Galanternik M., Jung HM., Pham VN., Weinstein BM. (2021).Live Imaging of Intracranial Lymphatics in the Zebrafish (2021). Circulation Researchorg/10.1161/CIRCRESAHA.120.317372
- Venero Galanternik M.*, Stratman AN.*, Weinstein BM. (2019). The Zebrafish cardiovascular system. The Zebrafish in Biomedical Research, American College of Laboratory Animal Medicine, 2020, Pages 131-143 – Book chapter. *These authors contributed equally to this work. doi.org/10.1016/B978-0-12-812431-4.00014-2.
- Santoro MM, Beltrame M, Panáková D, Siekmann AF, Tiso N, Venero Galanternik M, Jung HM and Weinstein BM (2019). Advantages and Challenges of Cardiovascular and Lymphatic Studies in Zebrafish Research. Cell Dev. Biol. 7:89. doi: 10.3389/fcell.2019.00089 – Meeting Review.
- Gore AV., Pillay*, Venero GalanternikM.*, Weinstein BM. (2018). The Zebrafish: A Fintastic Model for Hematopoietic Development and Disease. WIREs Dev Biol 2018 – Focus Review, WIREs Dev Biol 2018;e312 - Top 20 most read paper in Wiley Interdisciplinary Reviews - Developmental Biology
- Jung HM., Castranova D., Swift MR., Pham VN., Venero Galanternik M., Isogai, Butler MG., Mulligan TS., Weinstein BM (2017). Development of the larval lymphatic system in the zebrafish. Development doi: 10.1242/dev.145755
- Venero Galanternik M., Castranova D., Gore AV., Blewett NH., Jung HM., Stratman AN., Kirby MR., Iben J., Miller MF., Kawakami K., Maraia RJ., and Weinstein BM. (2017). A novel perivascular cell population in the zebrafish brain. eLife 2017;10.7554/eLife.24369
- Venero Galanternik M., Lush ME.,and Piotrowski T. (2016). Glypican4 regulates collective cell migration non-cell-autonomously. Dev Biol. 2016 Sep 15. pii: S0012-1606(16)30095-1. doi: 10.1016/j.ydbio.2016.09.002.
- Venero Galanternik M., Stratman, JungHM., ButlerMG. and Weinstein BM. (2016). Building the Drains: The Lymphatic Vasculature in Health and Disease - Advanced Review WIREs Dev Biol 2016. doi: 10.1002/wdev.246.
- Venero Galanternik M., Nikaido M., Yu Z., Mckinney AS. and Piotrowski T. (2016). Localized Gene Induction by Infrared-Mediated Heat Shock. Zebrafish, doi:10.1089/zeb.2015.1161.
- Venero Galanternik, M., Navajas Acedo, J., Romero-Carvajal, A., Piotrowski, T., (2016). Imaging collective cell migration and hair cell regeneration in the sensory lateral line. Methods Cell Biol 134, 211-256 - doi:10.1016/bs.mcb.2016.01.004
- Venero Galanternik, M., Kramer K. and Piotrowski T. (2015). Heparan Sulfate Proteoglycans Regulate Fgf Signaling and Cell Polarity during Collective Cell Migration, Cell Reports (2015), Cell Reports 10, 1–15
- Aguero, M. F., Venero, M., Brown, D. M., Smulson, M. E. and Espinoza, L. A. (2010). Phenoxodiol inhibits growth of metastatic prostate cancer cells. The Prostate, 70: 1211–1221. doi: 10.1002/pros.21156
- Nichol, P., Tyrrell, JD., Graham, B., Saund, R., Venero Galanternik, M., Cheng, S., Saijoh, Y. (2008). Short Article - Loss of Retinaldehyde dehydrogenase 2 expression during duodenal atresia formation in Fgf receptor 2IIIb null mice implicates a novel retinoic acid signaling pathway during duodenal development involving epithelial-mesenchymal interactions. Journal of the American College of Surgeons, ISSN: 10727515, Vol: 207, Issue: 3, pages: S57-S57
