Janis Weis
Professor of Microbiology and Immunology
Genetic Regulation of Lyme Disease

Molecular Biology Program
Education
B.A. University of Kansas
Ph.D. University of Minnesota Medical School
Research
We study the pathogenesis of Lyme disease; an infection caused by the tick-borne spirochete Borrelia burgdorferi that is responsible for 500,000 infections per year in the US. A spectrum of disease symptoms and severity are observed in infected patients, with arthritis being one of the most common. Using forward genetics we have identified host genes that regulate the severity of Lyme arthritis in inbred strains of mice. Upon infection with B. burgdorferi, C3H mice display severe disease while disease is mild in C57BL/6 mice. We have characterized numerous quantitative trait loci (QTL) that regulate the severity of Lyme arthritis. One of these, Bbaa2, identified the beta-glucuronidase gene (Gusb) as a major regulator of Lyme arthritis severity, with the defective allele resulting in accumulation of pro-arthritic glyclosaminoglycans in the joint tissue of infected mice. Gusb polymorphism also regulates the severity of an autoimmune rheumatoid arthritis model, suggesting variant alleles should be considered risk factors and could serve as targets for treatments for Lyme and rheumatoid arthritis patients.
A second QTL, Bbaa1, includes the Type I interferon (IFN) gene cluster, of particular interest as Type I IFN expression precedes the development of Lyme arthritis in joint tissues of C3H mice and is observed in Lyme disease patients. Positional cloning identified the Cdkn2a gene and its encoded protein, p19ARF, as the Bbaa1 regulator of IFNβ induced by B. burgdorferi. Both siRNA silencing and overexpression of p19ARF confirmed the association. p19ARF was found to act through parallel pathways involving the stabilization of the tumor suppressor p53 and sequestration of the transcriptional repressor BCL6. Our findings linking p19ARF regulation of p53 and BCL6 to the severity of IFNβ-induced Lyme arthritis in vivo reveal potential roles for p19ARF, p53, and BCL6 in Lyme disease and other IFN hyperproduction syndromes. Our studies further identified the muscle regulatory gene myostatin as a critical downstream mediator of Lyme arthritis development in mice, and point to additional therapeutic targets in arthritis development.
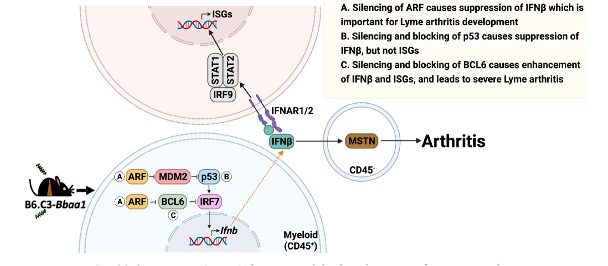
The heightened expression of the C3H allele of ARF in results in heightened expression of IFNβ and severe Lyme arthritis. ARF acts through p53 and BCL6 to upregulate IFNb in myeloid cells, resulting in in greater expression of the muscle regulatory protein myostatin in resident cells of the joint tissue. MSTN plays an unexpected role in Lyme arthritis development. 2022, PLoS Pathog 18(3): e1010365
We have also developed a model for one of the most troubling aspects of Lyme disease, the persistence of symptoms after what should have been an effective antibiotic regimen: post treatment Lyme disease syndrome (PTLDS). We have discovered that arthritis is sustained by bystander activation of CD4+ and CD8+ T lymphocytes. These T cells become a source of arthritis promoting IFNg, leading to inflammatory markers with striking similarity to those seen PTLDS patients. Bystander activated CD4+ and CD8+ bystander T cells from B. burgdorferi-infected mice display upregulated TLR2, which is central to their activation and promotion of arthritis. We are working to characterize bystander-activated T cells through single cell expression profiling, with the goal of identifying unique T cell markers as potential targets for resolution of arthritis.
Two additional Lyme arthritis models that exploit a dysregulated innate host response have been identified in collaboration with Ryan O’Connell’s lab: the miR146a-/- mouse and the miR-155-/- mouse. Previous studies had revealed the central role of the TLR2 adapter MyD88 and the NF-kB dependent transcriptional response in control of B. burgdorferi infection. Studies with miR146a-/- and miR-155-/- mice demonstrate that NF-kB dependent cytokines are also critical for the inflammatory responses leading to arthritis and carditis. These mice will provide models for testing novel therapeutics that reduce inflammation without impairing host defense.
Borrelia burgdorferi–infected mice expressing a single TCR transgene for a non–Borrelia epitope develop severe Lyme arthritis, revealing bystander activation of T cells is sufficient to promote disease. J. Immunol. 200: 1457–1470.
References
- Nat Rev Rheumatol. 2021 Aug;17(8):449-461. doi: 10.1038/s41584-021-00648-5. Epub 2021 Jul 5.PMID: 34226730
- Lochhead RB, Strle K , Arvikar SL , Weis JJ, Steere AC. Lyme arthritis: linking infection, inflammation and autoimmunity. 2021. Nature Rev Rheumatol. 8:449-461 PMID: 34226730
- Li J, Ma Y, Paquette,JK, Richards AC, Mulvey M, Zachary,JF, Teuscher C, and Weis JJ. The Cdkn2a gene product p19 alternative reading frame 9p19ARF) is a critical regulator of IFNb-mediated Lyme arthritis. 2022.PLoS Pathog 18(3): e1010365. https://doi.org/10.1371/journal.ppat.1010365. PMID: 35324997
- Whiteside, SK, Snook JP, Williams MA, Weis JJ. Bystander T cells: A balancing act of friends and foes. 2018, Trends in Immunology. 39:1021-1035
- Whiteside SK, Snook, JP, Ma Y, Sonderegger FL, Fisher C, Petersen, C, Zachary JF, Round JL, Williams MA, Weis JJ. IL-10 deficiency reveals a role for TLR2-dependent bystander activation of T cells in Lyme arthritis. 2018.J Immunology. 200:1457-1470 PMCID:PMC5809275
- Paquette JK, Ma Y, Fisher C, Li J, Lee SB, Zachary JF, Kim YS, Teuscher C, Weis JJ. Genetic Control of Lyme Arthritis by Borrelia burgdorferi Arthritis-Associated Locus 1 Is Dependent on Localized Differential Production of IFN-β and Requires Upregulation of Myostatin. 2017. J Immunology. 199:3525-3534. PCMID: PMC5679706
- Lochhead RB, Zachary JF, Dalla Rosa L, Ma Y, Weis JH, O'Connell RM, Weis JJ. Antagonistic Interplay between MicroRNA-155 and IL-10 during Lyme Carditis and Arthritis PLoS One. 2015 Aug 7;10(8):e0135142. doi: 10.1371/journal.pone.0135142. eCollection 2015. PMCID: PMC4529177
- Bramwell KK, Mock K, Ma Y, Weis JH, Zachary JF, Teuscher C, Weis JJ. beta-glucuronidase, a regulator of Lyme arthritis severity, modulates lysosomal trafficking and MMP-9 secretion in response to inflammatory stimuli. 2015. J Immunology 195:1647-56. PMCID: PMC4530054.
- Lochhead RB, Ma Y, Zachary JF, Baltimore D, Zhao JL, Weis JH, O’Connell RM, Weis JJ. MicroRNA-146a provides feedback regulation of Lyme arthritis but not carditis during infection with Borrelia burgdorferi. 2014. PLoS Pathog Jun 26;10(6):e1004212. PMCID: PMC4072785
- Bramwell KC, Ma Y, Weis JH, Chen X, Zachary JF, Teuscher C, Weis JJ. Lysosomal beta-glucuronidase regulates Lyme and rheumatoid arthritis severity. 2014. J Clin Invest 124:311-320. PMCID:PMC3871255
- Bramwell KC, Teuscher C, Weis JJ. Forward Genetics approaches for elucidation of novel regulators of Lyme arthritis severity. 2014. Front Cell Infect Microbiol eCollection 2014. PMCID:PMC4046100
- Lochhead* RB, Sonderegger* FL, Ma* Y, Brewster JE, Cornwall D, Maylor-Hagan H, Miller JC, Zachary JF, Weis JH, Weis JJ. Endothelial cells and fibroblasts amplify the arthritogenic Type I IFN response in murine Lyme disease and are major sources of chemokines in B. burgdorferi-infected joint tissue.2012. J Immunol 189:2488-2501. PMCID: PMC3424307
